- Blog
- Acer aspire one nav50 windows 7
- Daily nation newspaper nairobi today
- Masterwriter 2-0 mac serial number
- Malvern zetasizer equilibration time
- Head stone do not stand at my grave and weep
- Neverwinter nights platinum keys
- Capitan marvel ver
- Punch beat watch online dailymotion
- Delta wedding by eudora welty
- Jadwal kapal pelni pontianak
- Sengoku basara 2 op
- Windows 10 pro 64-bit product keys
- Dynomite deluxe serial
- Can deskcnc run hybrid stepper motor
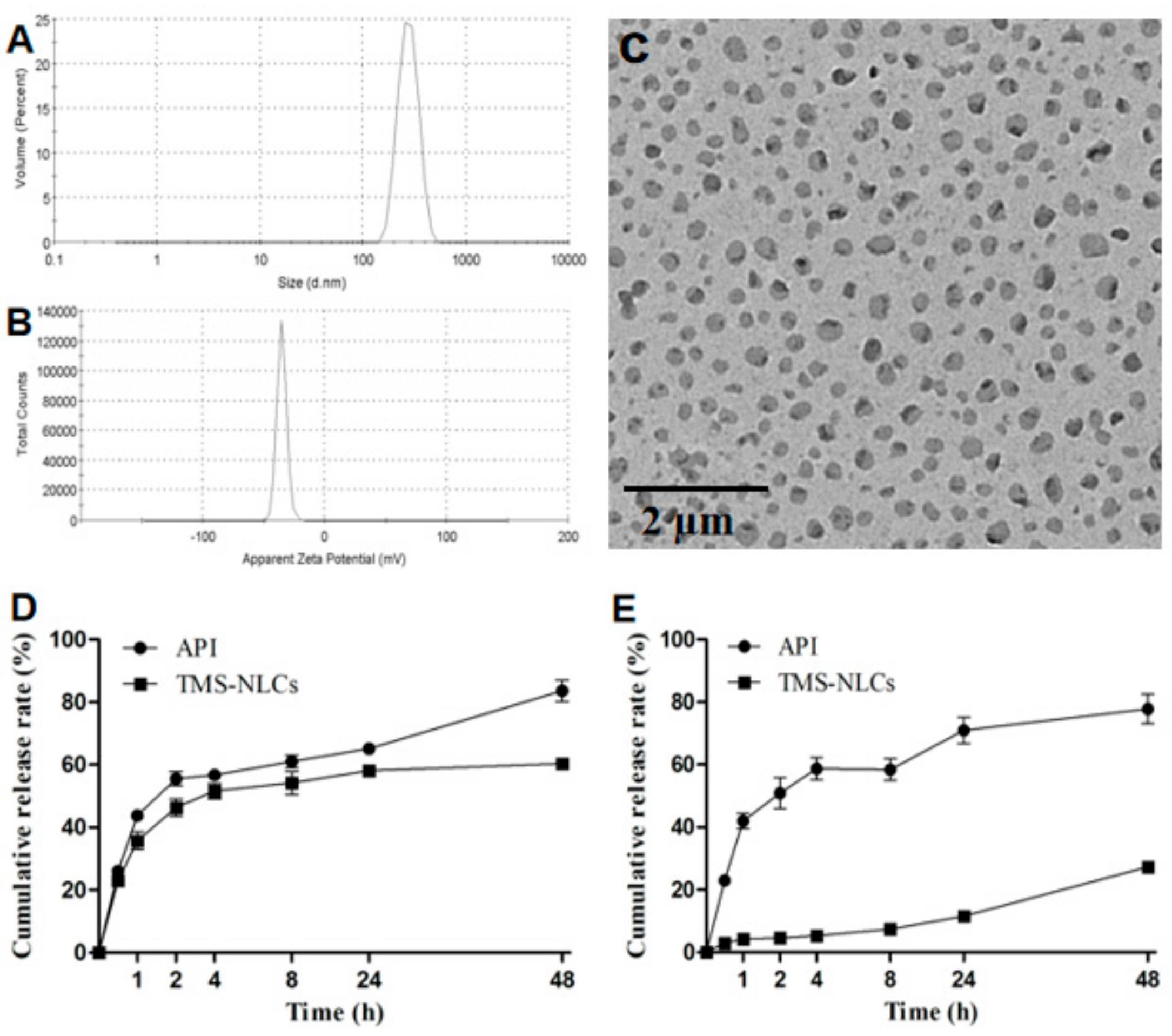
Nonetheless, a relevant downside to these techniques is their bias when characterizing polydispersed systems, due to their resolution being limited to a factor of 3, potentially failing to separate multimodal particle distributions. The combination of DLS and electrophoretic mobility (electrophoretic light scattering) further allows the estimation of the surface charge based on the zeta-potential distribution. Being fast and easy to use, DLS allows the estimation of size distribution and polydispersity index (PdI), which reflects the uniformity of a nanosystem.
.png)
The standard widely utilized characterization techniques are typically batch-mode analyses, such as dynamic light scattering (DLS). The carrier size, surface charge, and polydispersity (PdI) are the three major well-established properties known to affect the internalization and potentially the targeting of drug delivery systems within biological environments.

However, failure to ensure reliable and robust characterization, within in vitro settings, would directly impair the prediction of biological fate and limit success in in vivo settings. Therefore, by tailoring a nanocarrier’s features, we could augment its desired pharmacological effect. Changes in physicochemical properties of a nanocarrier can lead to a change in their behavior, as well as biological fate. The characterization of a nanosystem is a crucial initial step in the development of novel nanomedicine. However, many promises remain to be fulfilled, and recent years oversaw the trend of “back-to-basic”, trying to ensure a better understanding of the interplay between drugs, nanocarriers, and biological environment, especially biological barriers. Various types of nanoformulations (nanocarriers) have been proposed to impart biological superiority. Nanomedicine emerged as an advanced field expected to change the landscape of pharmaceutical development, promising improved drug efficacy and safety. QPM thus overcomes some of the drawbacks of the conventional methods, serving as a relevant complimentary technique in the characterization of nanosystems. Moreover, QPM full-field-of-view images enable the estimation of a single-particle-based size distribution, providing an alternative to the batch mode approach. Our results show the potential of QPM to localize subdiffraction-limited liposomes, estimate their size, and track their integrity over time. Image reconstruction was then performed with a phase-shifting algorithm providing high spatial resolution. A successful immobilization of liposomes in a non-dried setup allowed for static imaging conditions in an off-axis phase microscope. Liposomes were used as a model nanocarrier for their therapeutic relevance and structural versatility. In this study, we explore some of the downsides of batch-mode analyses and fluorescent labeling, while introducing quantitative phase microscopy (QPM) as a label-free complimentary characterization technique. Standard methods such as dynamic light scattering (DLS) and fluorescent-based assays present challenges in terms of system’s instability, machine sensitivity, and loss of tracking ability, among others. The rapid development of nanomedicine and drug delivery systems calls for new and effective characterization techniques that can accurately characterize both the properties and the behavior of nanosystems.
- Blog
- Acer aspire one nav50 windows 7
- Daily nation newspaper nairobi today
- Masterwriter 2-0 mac serial number
- Malvern zetasizer equilibration time
- Head stone do not stand at my grave and weep
- Neverwinter nights platinum keys
- Capitan marvel ver
- Punch beat watch online dailymotion
- Delta wedding by eudora welty
- Jadwal kapal pelni pontianak
- Sengoku basara 2 op
- Windows 10 pro 64-bit product keys
- Dynomite deluxe serial
- Can deskcnc run hybrid stepper motor
